Water Quality Lab
Partners: Mitchell and Michael
Pre-Lab Questions:
- Rain water is normally slightly acidic because carbon dioxide dissolves into it forming a weak carbonic acid, that results in the rain water having a pH of around 5.6. (Measuring Acid Rain)
- Oxygen-demanding wastes are pollutants because when there is a presence of large populations of bacteria that require oxygen, they deplete the dissolved oxygen levels in the water. This results in other organisms dying, which can disturb the natural food web in that ecosystem. (Water pollution FAQ Frequently Asked Questions)
- If the water quality of a water ecosystem is good, then the B.O.D. should be low causing the D.O. levels to be high. The opposite will occur if the water quality is poor.
- Sedimentation can occur in waterways in many ways, but the most frequent is run-off. If the topsoil is loose and it rains, then the topsoil will flush away into a nearby pond, which will increase the turbidity in the water.
- The impact of sedimentation on aquatic species can be harmful. Soil particles can cover important spawning beds for fish, aquatic insects, and plants. It can also increase the water temperature and will increase turbidity which reduces visibility for fish to prey on other organisms. Also not as much light will come through disturbing plant growth. (Soil In Our Streams)
- Warm aquatic temperatures on aquatic species can also be harmful because if the water temperature is too high then organisms will want to migrate north to cooler waters, but if it is blocked then many organisms will die. Also if the temperature is hot then dissolved oxygen levels will be low, making it very hard for organisms to survive.
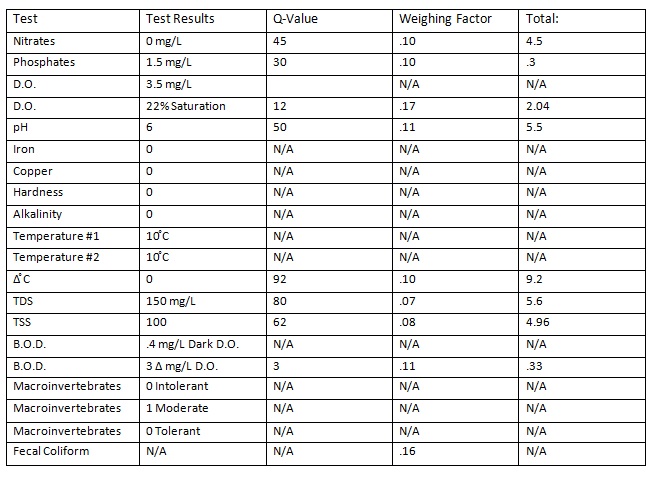
Data:
32.43 x 1.19 = 38.59
Water Quality Index (WQI) = 38.59 (Fair Water Quality)
Cummulative Index Value (CIV)
0 intolerant x 3 = o
1 moderate x 2 = 2
0 toleratnt x 1 = 0
Total CIV = 2 (Poor)
Water Quality Index (WQI) = 38.59 (Fair Water Quality)
Cummulative Index Value (CIV)
0 intolerant x 3 = o
1 moderate x 2 = 2
0 toleratnt x 1 = 0
Total CIV = 2 (Poor)
Post-Lab Questions:
- The water quality of the pond scored a fair water quality, falling between 26-50. In this case the WQI was 38.59. This means the water is not great and could be better but that could be from the turbidity levels being high. Also the levels of nitrates and phosphates were low.
- Of all the tests performed, the DO % saturation should have the greatest impact on the water quality rating because if the DO levels are low in an aquatic ecosystem, then it generally means that it is not very healthy. This is because if there is not enough oxygen in the water, then organisms will start to die or will migrate if possible to an area that has a higher DO level.
- If the Eco-Studies pond showed high nitrate levels, the source may be from a run-off of fertilizers that were spread around the school campus to grow the nearby plants and grass.
- The cause of high turbidity is usually from sedimentation that has been loosened by either deforestation or rain water. If the topsoil is loose, then it will move easily by wind or rain into nearby water. The increase of soil particles in the pond will increase the turbidity levels.
- In the data collected both sides were 10 degrees Celsius, but two causes that may change the temperature in the different sides of the pond are that one, there may have been more shade cover on one side of the pond from trees or buildings decreasing the side of one side and two, there may have been less vegetation on one side so the water absorbs the light instead of the plants increasing the temperature.
- A problem that might result from a higher pond temperature would be that dissolved oxygen levels will decrease making it harder for organisms like fish and plants to survive because all organisms rely on oxygen.
- Testing for fecal coliform bacteria was not important in this case because the pond is not being used for any human activities like using it for drinking, so the level of fecal coliform bacteria would not be needed.
- The other chemical tests that was collected was of iron, copper, hardness, and alkalinity. Copper and iron are measured in these tests to see what elements are in the water and are used to see if there any health concerns with the water. Alkalinity tests the waters ability to neutralize acids with bases. Hardness tests the calcium carbonate in the water. (Water Tests: What do the Numbers Mean?)
- Indicator species are used to assess water quality and are a good measure for determining water quality because indicator species are the first to die if the water quality is poor. They are a fast indicator of the water's quality. This helps scientists help treat the water quality. If the indicator species are thriving then that means the overall water quality is most likely good or excellent. The CIV number was less than 10 indicating that the water was poor, but the WQI number was between 26-50 indicating the water was fair. This may be due to the fact that the organisms in the collected area were frightened and moved away, so not many organisms were collected.
Bibliography:
Measuring Acid Rain. (n.d.). Retrieved December 10, 2015, from http://www3.epa.gov/acidrain/measure/index.html
Soil In Our Streams. (n.d.). Retrieved December 10, 2015, from http://www.bae.ncsu.edu/programs/extension/wqg/sri/sediment5.pdf
Water Tests: What Do the Numbers Mean? (n.d.). Retrieved December 10, 2015, from http://www.water-research.net/Waterlibrary/privatewell/whatdonumbers.pdf
Water Treatment Solutions. (n.d.). Retrieved December 10, 2015, from http://www.lenntech.com/water-pollution-faq.htm
Soil In Our Streams. (n.d.). Retrieved December 10, 2015, from http://www.bae.ncsu.edu/programs/extension/wqg/sri/sediment5.pdf
Water Tests: What Do the Numbers Mean? (n.d.). Retrieved December 10, 2015, from http://www.water-research.net/Waterlibrary/privatewell/whatdonumbers.pdf
Water Treatment Solutions. (n.d.). Retrieved December 10, 2015, from http://www.lenntech.com/water-pollution-faq.htm